Courtesy: Saqib Rehman MD
Director of Orthopaedic Trauma
Temple University
Philadelphia
Pennsylvania
USA
www.orthoclips.com
ALLOGRAFT & URIST STAGES OF ALLOGRAFT HEALING
History
• Jacob van Meekeren (1668 – Dutch) – first documented xenograft (from a dog) into skull of a soldier.
• Leopold Ollier’s “Traite de la regeneration des os” (1861) – first paper to formally define the term “bone graft”
• William MacEwen (1879 – Scottish) – first documented human allograft in a young boy.
• Vittorio Putti: Founder of orthopedic science, his paper in 1912 proposed principles of bone grafting.
• First full tissue banking in the world : The US Navy Tissue Bank (1949) in Bethesda
• First bone bank in India : Government General Hospital, Chennai (1997)
Bone graft
• Definition : Bone graft are bone that is transplanted from one area to another to aid in healing, strengthening or improving function.
• Bone or bone like material used in bone graft may come from
– Same person (Autograft)
– From a donor (Allograft)
– From another species (Xenograft)
– Man made source (Substitutes)
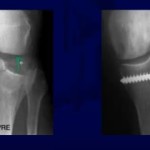
Bone Graft Indications
• To promote union in delayed union, non union, malunion, fresh fractures and osteotomies
• Void filler resulting from nonunion, cyst & tumors
• Bridge joints and perform arthrodesis
• Provide bone blocks to limit joint motion (arthroereisis)
• To help fusion between vertebrae, correct deformity & provide structural support.
Types Of Bone Graft
• Based on Source: Autograft, Allograft, Xenograft
• Based on contents: Cortical, Cortico-cancellous, Cancellous
• Based on Vascularity: Vascular, Non- Vascular
• Based on method of preservation: Fresh, Frozen, Freeze Dried, Demineralized
• Synthetic grafts or Substitutes
• Osteoinductive agents – rhBMP-2 (Infuse) and rhBMP-7 (OP-1)
• Orthopic Transfer: Host site is of same tissue as that from which graft was donated.
• Heterotopic transfer: To a new environment (eg- bone placed in a bed of soft tissue)
Properties Of Bone Graft
- Osteoinduction : Process of recruitment, proliferation & differentiation of host mesenchymal stem cells into chondroblast & osteoblast
- Osteoconduction : Process by which a graft acts as a scaffold passively hosting the necessary cells for healing
- Osteogenesis : Ability of a material to form new bone without the requirement of outside cells
Bone Graft Techniques
Onlay cortical grafts:
- Graft is placed subperiosteally across the fragments without mobilizing the fragments
- Usually supplemented with cancellous bone for osteogenesis
- Single and dual graft technique
- Most nonunion, malunion, Fixation, Arthrodesis etc
- Diaphyseal nonunions, the onlay technique is simpler and more efficient and has almost replaced the inlay graft
Inlay graft:
- A slot or rectangular defect is created in the cortex of the host bone
- A graft the same size or slightly smaller is fitted into the defect
- Occasionally used in ankle arthrodesis
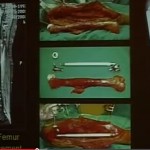
Autogenous Bone Graft
• From same individual
• “Gold standard” : Osteoconduction, Osteoinduction & Osteogenesis
• Drawbacks –
-Limited supply
-Donor site morbidity
Types
1. Cancellous
2. Cortical
3. Free vascular transfers
4. Muscle pedicle bone graft
5. Bone marrow aspirate
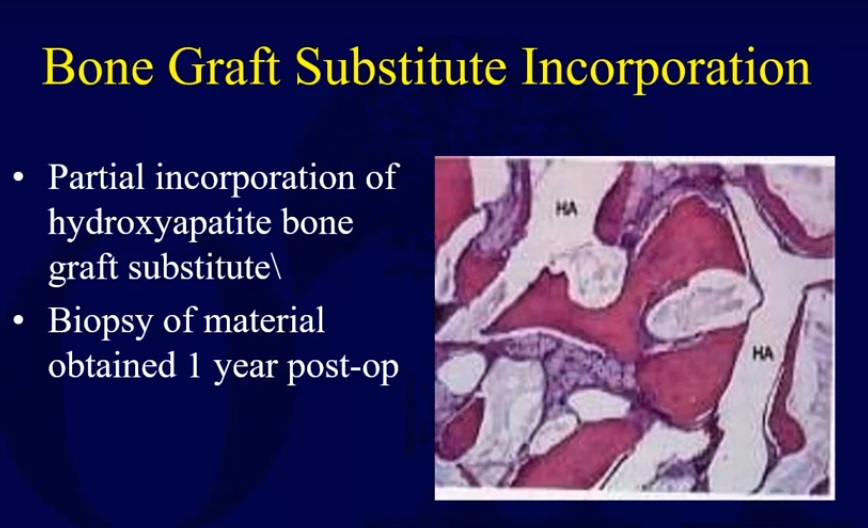
Bone Graft Substitutes
Allograft – Introduction
• Bone graft obtained from a cadaver and inserted after processing
• Properties
• Osteoconductive only due to lack of viable cells
• The degree of osteoconduction available depends on the processing method (fresh, frozen, or freeze-dried) & type of graft (cortical or cancellous)
• Advantages
-Unlimited supply
-Lack of donor site morbidity
• Disadvantages
-Lack of osteoinductive and osteogenic properties
-Risk of disease transmission
-Reduced mechanical properties from processing
Bone Bank
• To provide safe and useful allograft materials efficiently
• Donor must be screened for bacterial, viral, fungal infections
• Contraindication to donation : Malignancy, collagen vascular disease, metabolic bone disease and the presence of toxins
• Bone & ligament and bone & tendon are banked now
• Articular cartilage and menisci can be cryopreserved
Preperation Of Allograft
• Chloroform – methanol is used to extract lipids and cell membrane lipoproteins
• Hydrochloric acid extracts soluble proteins & requires 24 hours to demineralize the surface
• Sterilization also done with irradiation & ethylene oxide (kills bacteria and viruses)
• Neutral phosphate buffer is used to remove endogenous intracellular and extracellular transplantation antigen
• The bone is then frozen and freeze dried and stored at -70 to -80?C
Processing methods
• Fresh allograft
– Cleansing and processing removes cells and decreases the immune response improving incorporation
– Rarely used due to disease transmission and immune response of recipient
• Frozen or freeze-dried
-Reduces immunogenicity while maintaining osteoconductive properties
-Reduces osteoinductive capabilities
• Shelf life
– Two years for fresh frozen stored at -20?C
– Five years for fresh frozen stored at -70?C
– Indefinite for freeze-dried
Risks & Complications of Bone grafting
Disease Transmission
- Hepatitis B – risk in musculoskeletal fresh-frozen allograft transplantation is 1 in 63,000
- Hepatitis C – risk in musculoskeletal fresh-frozen allograft transplantation is 1 in 100,000
- HIV – risk of transmission in fresh-frozen allograft bone is 1 in 1,000,000 – 1,670,000
- Allografts are tested for HIV, HBV, HCV, HTLV-1, and syphilis
Serous wound drainage
- Calcium sulfate bone graft substitute associated with increased serous wound drainage
Stages Of Bone Graft Healing – Autograft
URIST STAGES OF ALLOGRAFT HEALING
1. Inflammation
2. Revascularization and Cellular Infiltration
3. Osteoinduction
4. Osteoconduction
5. Remodeling and Incorporation
Stage I – Inflammation
- Timeline: Immediate to Day 3
- Key Features:
– Graft implantation causes local tissue injury.
– Neutrophils and macrophages dominate early response.
– Macrophages phagocytose debris and secrete cytokines (IL-1, IL-6, TNF-?).
– Initiates cascade recruiting mesenchymal stem cells (MSCs).
-Release of damage-associated molecular patterns (DAMPs). - Clinical Relevance
-Avoid excessive debridement to preserve early cytokine signaling.
-NSAID use should be balanced to avoid suppressing osteogenesis.
• Histologic Insight – Inflammation
- H&E staining shows neutrophilic infiltration.
- Necrotic bone margins stimulate cytokine release.
- Histological evidence of micro-vascular damage.
- Immunostaining for CD68 shows macrophage localization.
Stage II – Revascularization and Cellular Infiltration
• Timeline: Days 3–14
• Key Events:
– Vascular endothelial growth factor (VEGF) upregulated by hypoxia.
– Capillaries sprout from host bed into graft.
– MSCs, fibroblasts, and endothelial cells infiltrate graft scaffold.
– Essential for nutrient delivery and waste removal.
• Clinical Relevance:
-Avoid compressive graft fixation that limits blood supply.
-Autologous bone marrow aspirate can enhance MSC availability.
• Histologic Insight – Revascularization
-CD31+ staining confirms new vessel formation.
-Graft bone shows early sinusoidal invasion.
-Angiogenesis correlates with BMP expression.
– Macrophage polarization (M2 type) supports regeneration.
Stage III – Osteoinduction
• Timeline: Days 7–21
• Definition:
– Biochemical stimulation of MSCs to differentiate into osteoblasts.
– Governed primarily by bone morphogenetic proteins (BMPs)
• Biological Events:
-BMPs bind to receptors on MSCs.
– Activation of SMAD signaling pathways.
-Osteogenic transcription factors: RUNX2, Osterix.
• Osteoinduction – Key Molecules
-BMP-2, BMP-7 – essential for osteoblast differentiation.
– TGF-?, PDGF, IGF-1 – secondary regulators of osteogenesis.
– Runx2 – initiates osteoblast gene expression.
– ALP (alkaline phosphatase) – early marker of osteoblast activity.
• Clinical Applications:
– Demineralized bone matrix (DBM) retains osteoinductive proteins.
– Recombinant BMPs (rhBMP-2/7) used in spine fusion and tibial nonunion.
Stage IV – Osteoconduction
• Timeline: Days 14 – 42
• Definition: The passive process of new bone growing along the scaffold of the graft.
• Histologic Events:
-Osteoblasts line existing trabeculae.
– Woven bone deposition observed on graft surfaces.
– Host cells colonize necrotic graft matrix.
• Clinical Considerations:
-Structural allografts provide excellent scaffolding but poor induction.
– Graft preparation (freeze-drying, irradiation) affects osteoconduction.
• Osteoconduction – Scaffold Properties
– Porosity: Must permit cell migration and vascular in-growth.
– Surface topography: Enhances cell attachment (e.g., rough surfaces).
– Biodegradability: Supports replacement by viable host bone.
• Materials Used: Cortical/cancellous allografts & Hydroxyapatite or beta-TCP coatings.
Stage V – Remodeling and Incorporation
• Timeline: Weeks to Months (can last up to 12–18 months)
• Biological Sequence:
– Osteoclast-mediated resorption of necrotic bone.
– Osteoblast-mediated deposition of lamellar bone.
– Formation of new Haversian systems.
•Outcome:
-Functional union with host bone.
-Structural and biomechanical integrity restored.
• Clinical Correlates:
– Failure in remodeling results in fibrous union or graft failure.
– In immunocompromised or elderly, this phase is often delayed.
• Histologic Features – Remodeling
-TRAP+ staining: Active osteoclasts resorbing dead bone.
-Dynamic labeling with tetracycline shows new bone deposition.
– Full incorporation leads to indistinguishable graft-host boundary.
Radiologic Correlation of Stages
• Stage I–II: Radiolucency around graft edges; soft tissue swelling.
• Stage III–IV: Early mineralization and bridging trabeculae.
• Stage V: Homogeneous bone density; cortical continuity.
Factors Influencing Healing Across URIST Stages
• Host Immunity – Excessive response can reject graft.
• Graft Type – Fresh-frozen vs freeze-dried; cortical vs cancellous.
• Graft Size – Larger grafts require more time for revascularization.
• Mechanical Environment – Stability is crucial; micromotion impairs integration.
• Comorbidities – Diabetes, smoking, and steroids delay healing.
Enhancing Allograft Healing
• Use of bone marrow aspirate concentrate (BMAC).
• Platelet-rich plasma (PRP) – source of growth factors.
• Local delivery of rhBMPs.
• 3D-printed scaffolds with custom porosity.
• Gene-activated matrices delivering osteogenic factors.
Clinical Applications of the URIST Model
• Spine Surgery – Interbody fusion with DBM and BMP.
• Revision Arthroplasty – Femoral or acetabular bone loss.
• Tumor Resection – Large segmental grafting.
• Nonunions – Augmentation with osteoinductive agents.
Complications in Graft Healing
• Nonunion or fibrous union.
• Graft rejection or immune reaction.
• Infection – especially with structural allografts.
• Resorption without remodeling.
• Mechanical failure – fracture of graft segment

Leave a Reply